Ever bought a generic drug in the U.S. and then seen the same pill for half the price in Canada or India? It’s not a trick. It’s the reality of how generic drugs work across the world. The same active ingredient, same dosage, same manufacturer - but wildly different prices, availability, and even quality depending on where you live.
How Much of the World Uses Generic Drugs?
Global use of generic drugs ranges from 50% to 90% of all prescriptions, but that number hides a deep divide. In the United Kingdom, 83% of all prescriptions are filled with generics. In Germany, it’s 80%. In the Netherlands, 70%. Meanwhile, in Switzerland, only 17% of prescriptions are for generics. Why? It’s not about need - it’s about policy, pricing, and culture.
The U.S. leads in volume: over 90% of prescriptions are for generics. But here’s the twist - Americans pay more for those generics than almost any other country. In 2022, U.S. drug prices (including brand and generic) were 2.78 times higher than the average of other wealthy nations. So why do we use so many generics and still pay so much?
Why Do Prices Differ So Much?
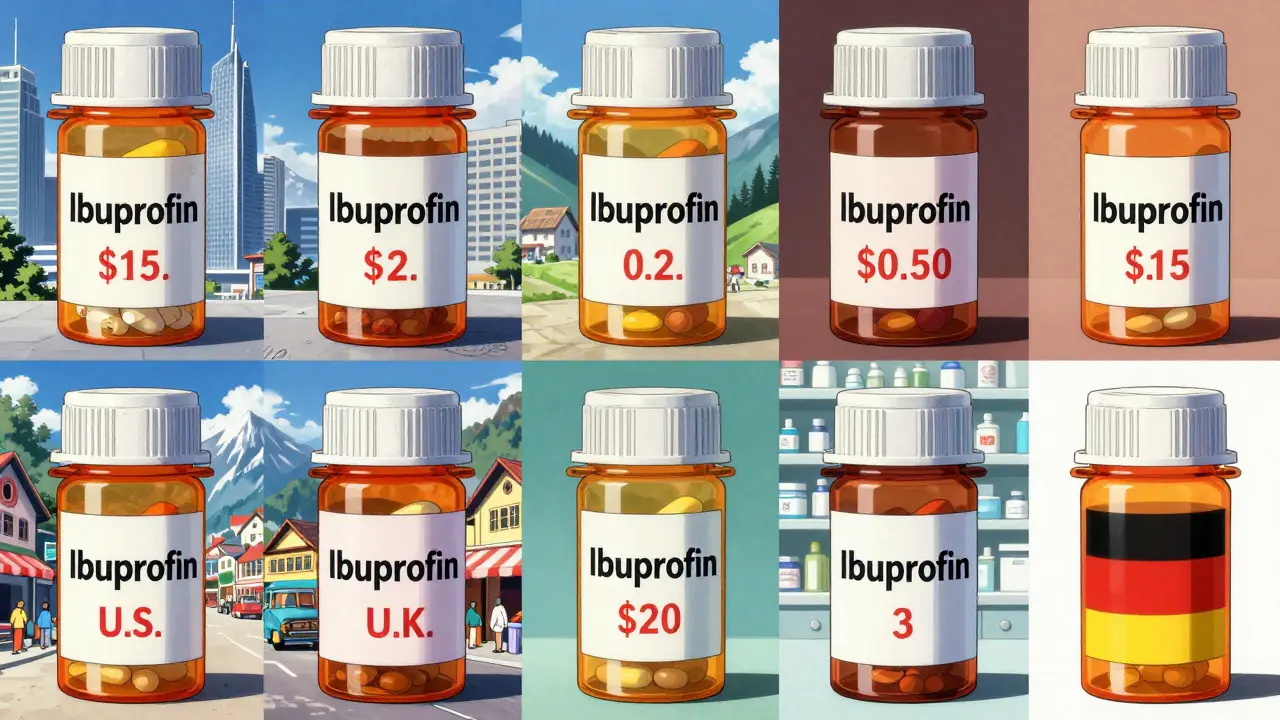
Imagine buying the same bottle of ibuprofen in five countries. In the U.S., it might cost $15. In the U.K., $2. In India, 50 cents. In Switzerland, $20. What’s going on?
It starts with competition. In markets like the U.S. and the U.K., dozens of companies can make the same generic drug. The U.S. has two or more manufacturers for 66% of off-patent drugs. The U.K. has 50%. But in countries like Switzerland and Italy, only one or two companies are allowed to sell - often because regulators delay approvals or don’t push for substitution. Less competition means higher prices.
Then there’s reimbursement. In Germany, if you prescribe a generic, the pharmacy gets paid the same as if you prescribed the brand. That encourages doctors to choose generics. In Switzerland, doctors get paid more if they prescribe the brand. So they do.
And then there’s parallel trade. When prices differ by 300%, people start buying across borders. Canadians import U.S. drugs. Americans order from Canadian pharmacies. Germans buy from Poland. It’s legal in some places, illegal in others - but it’s happening.
Who Makes the World’s Generic Drugs?
India produces about 20% of the world’s generic drugs. It supplies 40% of the generics used in the U.S. That’s why so many pills in your medicine cabinet say "Made in India."
But quality isn’t uniform. A 2023 study from Ohio State University found that generics made in India had a 54% higher rate of severe adverse events - including hospitalizations and deaths - compared to identical drugs made in the U.S. The issue isn’t the active ingredient. It’s the fillers, the coating, the manufacturing environment. A pill made in a facility with poor temperature control or dirty equipment can behave differently in your body.
China is catching up fast. Its FDA-approved manufacturing facilities jumped from 12 in 2010 to 187 in 2023. But inspection reports show similar quality gaps. The FDA inspects foreign plants - but often gives advance notice. That lets factories clean up before inspectors arrive. In the U.S., inspections are unannounced. That’s why U.S.-made generics are often more reliable, even if they cost more.

Why Do Some Countries Resist Generics?
It’s not always about money. In Switzerland and Italy, doctors and patients trust the brand name more. They believe it’s safer. Even when generics are cheaper and approved by regulators, people stick with what they know.
Regulatory systems also slow things down. In Europe, a generic drug can get approval from the European Medicines Agency - but then must be approved individually in each country. That adds 18 to 24 months to market entry. In the U.S., once the FDA approves, it’s approved everywhere. No delays. No red tape.
And then there’s patent manipulation. Drug companies file minor changes - a new coating, a different pill shape - to extend their monopoly. Between 2015 and 2022, 1,247 such "evergreening" patents were filed on just 12 top-selling drugs. These aren’t new medicines. They’re legal tricks to block generics.
What Happens When Generics Don’t Work?
Some patients notice real differences. One Reddit user reported switching from a U.S.-made generic levothyroxine to an Indian-made version - and suddenly felt fatigued, gained weight, and had heart palpitations. Their doctor had to switch them back.
That’s not rare. Pharmacists on forums like the American Medical Association’s physician network report patients coming in with unexpected side effects after switching generics - especially when they travel or refill prescriptions online. The active ingredient is the same. But the way it’s absorbed? That can vary.
For drugs with narrow therapeutic windows - like blood thinners, seizure meds, or thyroid hormones - even small differences matter. The FDA allows generics to be 80-125% as effective as the brand. That’s a 45% range. For some people, that’s enough to feel worse.

What’s Changing Now?
The U.S. Inflation Reduction Act of 2022 is pushing the FDA to speed up generic reviews by 30%. It’s also funding more unannounced inspections of foreign factories.
Europe is trying to harmonize. The European Commission wants 80% generic use across all member states by 2030. They’re pushing for automatic substitution - meaning pharmacists can switch you to a generic without asking your doctor.
India’s market is growing. It hit $28 billion in 2024 and is expected to reach $51 billion by 2033. But with growth comes pressure. Manufacturers cut corners to stay competitive. The FDA is stepping up inspections - but it’s a race against time.
And then there’s AI. By 2030, machine learning could cut generic development time from 3-5 years to 18-24 months. That means faster access - but only if regulators keep up.
What Should You Do?
If you’re on a generic drug:
- Stick with the same manufacturer if it works for you. Don’t switch unless your pharmacist or doctor says it’s safe.
- Check the pill’s imprint code. If it changes, ask why.
- For critical meds - thyroid, epilepsy, blood thinners - ask your pharmacist if the generic is bioequivalent to your previous version.
- If you’re traveling, bring your prescription and know your drug’s name in the local language. Your U.S. generic might not exist abroad.
And if you’re shopping for meds online: be cautious. A 60% price drop might mean a 100% risk drop in quality.
Why are generic drugs cheaper than brand-name drugs?
Generic drugs are cheaper because they don’t need to repeat expensive clinical trials. Once a brand-name drug’s patent expires, other companies can make the same medicine using the existing safety data. They only need to prove bioequivalence - that their version works the same way in the body. That cuts development costs by 80-90%, which lets them sell at lower prices.
Are generic drugs as safe as brand-name drugs?
Generally, yes - but not always. The FDA and EMA require generics to meet strict standards. However, quality depends heavily on manufacturing. A generic made in a facility with poor controls can have inconsistent absorption or harmful contaminants. Studies show higher adverse event rates for generics made in certain countries, especially for older drugs where cost pressures are highest.
Can I trust generics made in India or China?
Many are safe - millions of people use them every day. But quality varies. The U.S. FDA inspects foreign factories, but often gives advance notice, which lets companies prepare. U.S.-made generics undergo unannounced inspections, making them more reliable. If you’re on a critical medication, ask your pharmacist which facility made your pill. Some manufacturers have better track records than others.
Why do some countries use more generics than others?
It’s a mix of policy, culture, and economics. Countries like the U.K. and Germany have laws that require or strongly encourage pharmacists to substitute generics. They also reimburse generics at the same rate as brands. In places like Switzerland and Italy, doctors and patients prefer brand names, and reimbursement rules favor them. In the U.S., high volume comes from pharmacy laws that allow automatic substitution - but prices remain high due to market consolidation and lack of price controls.
Do generic drugs cause more side effects?
Not inherently. But differences in inactive ingredients - fillers, dyes, coatings - can affect how a drug is absorbed. For some people, especially those on thyroid, epilepsy, or blood thinner meds, switching generics can trigger side effects. That’s why it’s important to stay on the same manufacturer if your body responds well to it. Always report changes in how you feel after a switch.
Is it legal to buy generic drugs from other countries?
In the U.S., importing prescription drugs from other countries is technically illegal, though enforcement is rare for personal use. Many people buy from Canadian pharmacies because prices are lower and quality is generally high. But buying from unregulated online sellers - especially from India or Southeast Asia - carries risk. You might get a counterfeit, expired, or contaminated product. Stick to licensed pharmacies with verified credentials.
Final Thoughts
The global generic drug market isn’t broken - it’s fragmented. The same pill can be made in three different countries, sold at six different prices, and regulated under five different rules. What’s consistent? The need for affordable medicine. But affordability doesn’t mean equal access. It means navigating a patchwork of policies, manufacturers, and quality controls.
The future of generics depends on two things: better global standards and smarter regulation. Until then, patients need to be their own advocates - know your drug, know your source, and don’t assume "generic" means "identical."

Shalini Gautam
February 24, 2026 AT 08:51As an Indian, I’m proud of what our pharma industry has done for the world. We make over 40% of the U.S.’s generics - and yes, some of it’s cheap, but that’s because we’re efficient, not negligent. Factories in Gujarat and Hyderabad follow WHO-GMP standards. The FDA finds violations? Sure, but they find them in U.S. plants too. We’re not the problem - profit-driven monopolies are.
Stop demonizing Indian-made meds. My cousin takes blood pressure pills made in Mumbai. She’s been stable for 7 years. Meanwhile, my cousin in Ohio pays $80 for the same thing. That’s not a quality issue - that’s a greed issue.
Let’s be real: if the U.S. allowed price negotiation like Canada or the U.K., we wouldn’t be having this conversation. We’d be celebrating how affordable life-saving drugs are. Instead, we’re scared of pills because Big Pharma told us to be.
Timothy Haroutunian
February 24, 2026 AT 13:50Look, I get that generics are cheaper, but let’s not pretend they’re all the same. I switched from my brand-name levothyroxine to a generic made in India last year - and I felt like a zombie for three months. Weight gain, brain fog, heart palpitations. My endocrinologist had to switch me back. Turns out, the inactive ingredients - the dyes, the binders - they’re not regulated the same way. The FDA allows up to a 45% variation in absorption. That’s not a bug - that’s a feature of the system.
And don’t get me started on how the FDA inspects foreign plants. They give advance notice. I’m sorry, but if you’re going to inspect a facility that’s making medicine for millions of Americans, you don’t call ahead. You show up unannounced like you do in the U.S. Otherwise, you’re just playing dress-up with compliance.
So yeah, I use generics. But I stick to the same manufacturer. I check the imprint code. I know the pill looks different? I ask. Because I’m not a lab rat for corporate cost-cutting.
And if you’re buying pills off some shady website because it’s $5 instead of $50? You’re not saving money. You’re gambling with your life. And that’s not brave - it’s stupid.
Ashley Johnson
February 25, 2026 AT 06:44EVERYTHING IS A LIE. The FDA? They’re in bed with Big Pharma. You think they inspect plants? No. They get paid by the companies they inspect. The pills you take? Half of them have trace amounts of carcinogens. They call it ‘acceptable levels’ - but what’s acceptable? Your death? Your child’s death?
India? China? They’re dumping toxic waste into the water and then making pills out of it. The ‘active ingredients’ are real - but the fillers? Heavy metals. Mold. Rat hair. I’ve seen the reports. The FDA doesn’t publish them. Why? Because they’re scared.
And don’t even get me started on the ‘bioequivalence’ nonsense. 80-125%? That’s not medicine. That’s a roulette wheel. One day you feel fine. Next day, you’re in the ER. That’s not a coincidence. That’s design.
They want you to think you’re saving money. But you’re not. You’re buying poison. And they’re laughing all the way to the bank.
tia novialiswati
February 26, 2026 AT 15:38Hey everyone - I just want to say: if you’re on a critical med like thyroid or seizure meds, DON’T SWITCH WITHOUT TALKING TO YOUR PHARMACIST. Seriously. I switched generics once because it was cheaper - and I had a panic attack for 48 hours. Turned out the new version had a different coating that changed how fast it dissolved. Scary stuff.
But here’s the good news: your pharmacist can tell you which manufacturer made your pill. Ask for the lot number. Look it up. Some companies have way better track records. And if you’re traveling? Bring your meds. Don’t trust foreign pharmacies unless they’re licensed.
You got this. You’re not alone. And you deserve safe, affordable medicine. Stay informed. Stay calm. And always ask questions. 💪
Lillian Knezek
February 28, 2026 AT 07:24They’re injecting us with Chinese nanobots through the pills. That’s why the side effects are so weird. The FDA knows. They just won’t say it. The ‘fillers’? They’re tracking devices. The ‘coatings’? They’re signal transmitters. I read the patent filings. It’s all there. You think your thyroid med is helping you? It’s controlling you.
And don’t get me started on the 5G towers. They sync with the pill’s metallic coating. That’s why people get dizzy after switching brands. It’s not chemistry. It’s surveillance.
Check your pill under UV light. You’ll see the code. I did. I saw ‘X3-7’ - that’s the tracking ID. I’m not crazy. I’m awake.
Christopher Brown
March 2, 2026 AT 03:18U.S. generics cost more because we don’t have price controls. Simple. India makes them cheap. Canada negotiates. We? We let monopolies rip us off. The solution? Public procurement. Like the VA does. Stop pretending it’s about quality. It’s about power.
Khaya Street
March 4, 2026 AT 01:19As someone from South Africa, I’ve seen firsthand how access to affordable generics saves lives. In rural clinics, we rely on Indian-made antiretrovirals. No one’s complaining about the taste - they’re alive. Quality control isn’t perfect, but neither is our healthcare funding. We prioritize access over ideology.
Westerners act like generics are a gamble. But for billions, they’re the only choice. Maybe we should ask: why do we have to choose between affordability and safety? It shouldn’t be that hard.
Christina VanOsdol
March 6, 2026 AT 00:35Let’s cut through the noise: the real scandal isn’t Indian factories - it’s the U.S. pharmacy benefit managers (PBMs). They’re the middlemen who negotiate drug prices - and they pocket the difference. They push generics that give them the highest rebate - not the best quality. So you get a pill from a factory with 30% contamination risk… because it’s profitable.
The FDA inspects? Sure. But they’re underfunded. And the ‘unannounced’ inspections? Often scheduled 3 days in advance. The companies clean up. The inspectors see a show. The public gets a placebo of safety.
And don’t forget: 80% of the active ingredients in U.S. generics come from China. That’s not a supply chain - it’s a vulnerability. We’re one pandemic, one geopolitical rift, one factory shutdown away from a national crisis. And we’re still letting PBMs decide what you take.
It’s not about where it’s made. It’s about who’s profiting from your dependence.
Brooke Exley
March 6, 2026 AT 12:04Y’all are overthinking this. If your drug works - stick with it. If it doesn’t - talk to your pharmacist. Period.
I’m a nurse. I’ve seen people switch generics because they’re ‘cheaper’ - and then panic when they feel off. But guess what? 90% of the time, it’s not the pill. It’s stress. It’s sleep. It’s dehydration. Or they just didn’t take it on an empty stomach like they’re supposed to.
That said - if you’re on a narrow-window drug? Don’t switch unless you have to. Ask for the manufacturer. Write it down. Keep the same one. And if your pharmacist says ‘it’s bioequivalent’ - trust them. They’re the experts.
Don’t let fear drive your health decisions. Knowledge does. And you’ve got this. 💖
Nick Hamby
March 8, 2026 AT 04:48The deeper question here isn’t about manufacturing standards - it’s about epistemic justice. Who gets to define ‘quality’? Who gets to decide what ‘safe’ means? In the U.S., we’ve outsourced not just production, but also trust. We’ve ceded authority to corporate entities, regulatory agencies, and global supply chains - while the patient remains an afterthought.
Generics are not merely pharmaceutical products. They are social contracts. The fact that a pill made in Hyderabad can be legally sold in Ohio at 1/30th the price - yet deemed ‘equivalent’ - reveals a fundamental asymmetry in how value is assigned. Is it the molecule? Or the market? Or the moral calculus of who gets to live?
Perhaps the real crisis isn’t inconsistent fillers - it’s that we’ve normalized inequality as a feature of medicine. And until we treat access to health as a human right - not a commodity - we’ll keep debating pills while the system keeps selling them.